Master Every Aspect of the Medical Device Design & Development With Taining
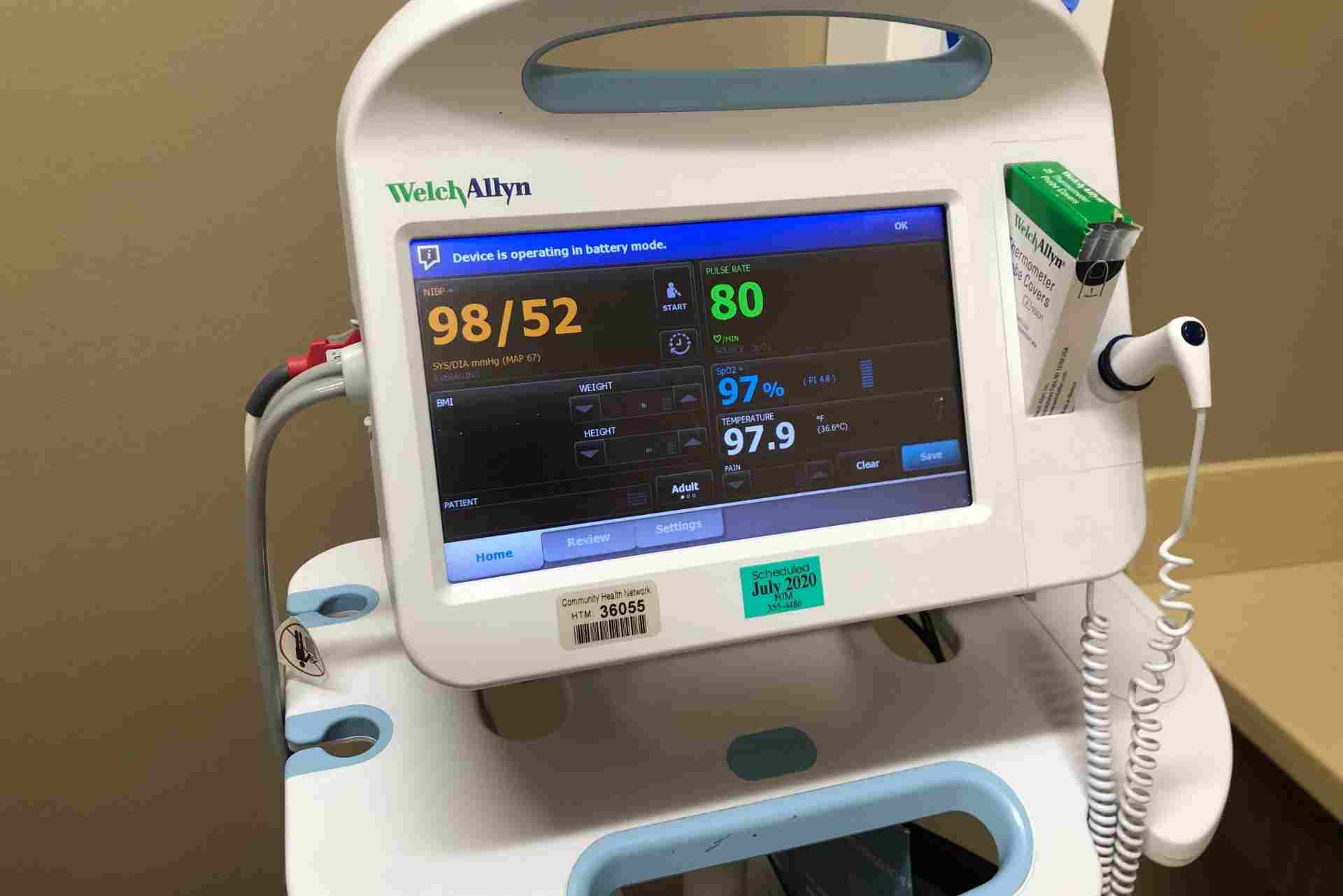
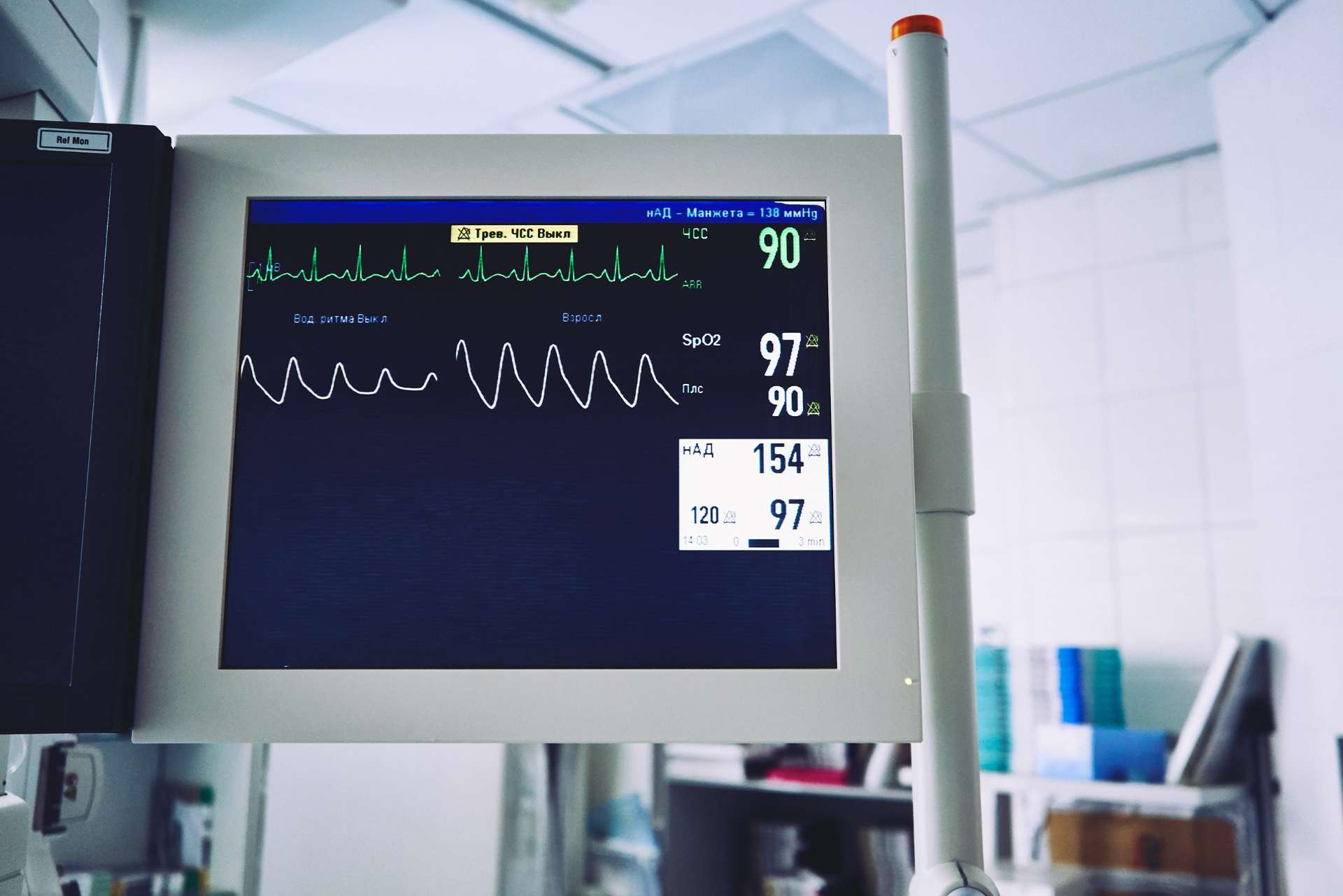
When designing and developing a new medical device, quality and patient safety must be at the forefront of all processes, inputs, studies and more. For companies with legacy products under the old MDD, article 10 of the Medical Device Regulation (MDR) requires the design of all products which can be a challenge.
We have a range of courses that support the design and development of a medical device which are listed below.
Team Training Solutions
We support medical device regulatory leaders improve the technical skills and knowledge of their employees, so they are equipped to overcome challenges of the MDR and meet business objectives. Working together, we design and deliver technical training courses that align with your learning requirements and business goals. For more information about how we can support your team please get in touch.
Could your team benefit from any of the courses listed above? Or maybe a topic which is not highlighted? We would be happy to discuss your objectives and see how we might be able to help.